- Blog
- Mac control shift eject not working
- Jesus culture dance with me lyrics
- Mathematica 11-2 crack
- Apache air assault pc torrent
- Radio motorola mag one a8 mercadolibre
- Disable community blocksworld
- Download kingdom hearts 2 final mix iso
- Backstreet boys i want it that way composer
- Fifa 16 redbox
- Octopus box lg tool crack
- Automation studio 2-0
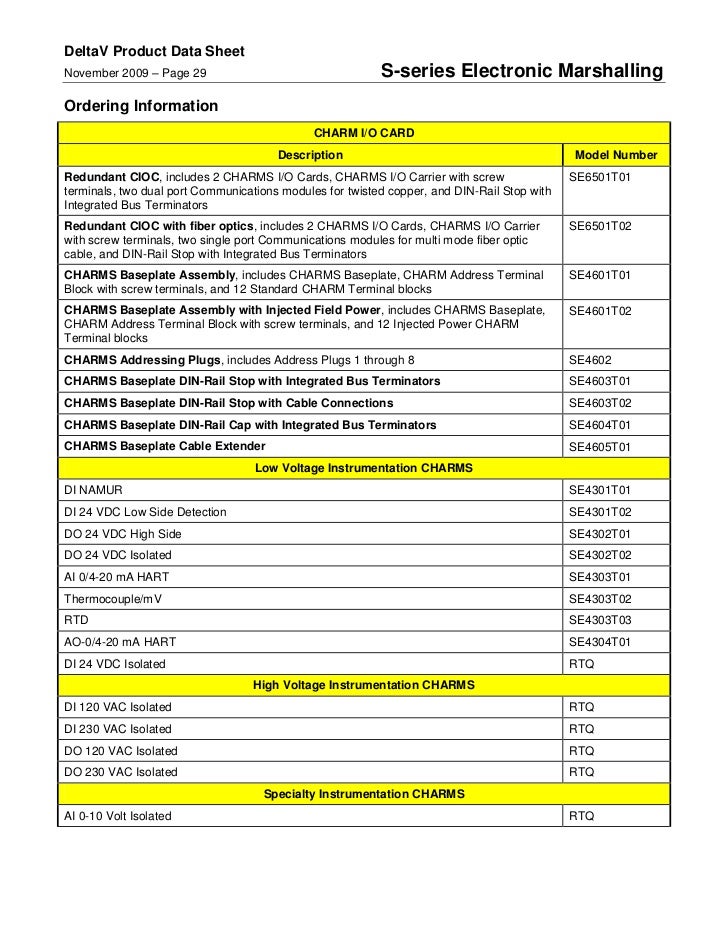
- Which delta v charm to use with micro motion pulse input
- Divinity original sin 2 nude mod
- English grammar modal verbs exercises
- Mini vci for toyota tis techstream
- Solidworks 2010 service pack 1
- Best form of magnesium for muscle relaxation
- Free video enhancer online
- Sten mark 2 9 mm
- Docker remove container silent
- Polybius square decoder

To complete the cooling process, we don't just illuminate the gas from one direction, but from six-the same color of light is shot into each side of a cube. This happens to put more atoms into the velocity range where they can absorb light and be slowed, which keeps the cooling process going. After we have slowed a small fraction of atoms, they will collide with their neighbors, redistributing the remaining energy among the atoms in the gas. The atoms are always colliding with each other, which is what keeps a gas in thermal equilibrium. If we choose our light color so that only atoms moving towards the light source absorb light, then the net result is that the atoms will be slowed until they stop absorbing light.Ĭollisions also play their part. The absorbed photon always gives the atom a kick in the direction of the light beam, while the other photon is emitted in a random direction. These atoms each absorb a photon and then emit a photon. The sharp definition of the absorption lines and the Doppler shift lets us choose a color of light that is only absorbed by the fraction of atoms moving at a particular speed relative to a light beam. However, the motion of the atoms brings the Doppler shift into play, meaning that the color of the light actually absorbed by a particular atom depends on its relative motion. Dilute atomic gases typically absorb light at a particular set of sharply defined colors. To understand the complexity of the problem, let's compare the results of standard optical cooling on atoms and molecules. This means that, in ideal conditions, reducing the rotation of one thing must result in something else beginning to rotate. Furthermore, rotational motion involves angular momentum, which is a conserved quantity. For a molecule, there is a temperature associated with the translational motion, a temperature associated with the internal motions of the molecule, and a combined temperature. For an atom, temperature is usually just related to translational motion. Why does this matter? Because an object's temperature is basically a sum of its kinetic energy. However, molecules are a different story because the atoms that make up the molecule can vibrate and rotate.

For atoms, this is a relatively straightforward process (note that straightforward is not the same as simple) because atoms have only a couple of degrees of freedom, the most important of which are translational motion and the electronic state. The ability to cool stuff down to micro- and nanokelvin temperatures has become a significant tool in the exploration of quantum physics.
- Blog
- Mac control shift eject not working
- Jesus culture dance with me lyrics
- Mathematica 11-2 crack
- Apache air assault pc torrent
- Radio motorola mag one a8 mercadolibre
- Disable community blocksworld
- Download kingdom hearts 2 final mix iso
- Backstreet boys i want it that way composer
- Fifa 16 redbox
- Octopus box lg tool crack
- Automation studio 2-0
- Which delta v charm to use with micro motion pulse input
- Divinity original sin 2 nude mod
- English grammar modal verbs exercises
- Mini vci for toyota tis techstream
- Solidworks 2010 service pack 1
- Best form of magnesium for muscle relaxation
- Free video enhancer online
- Sten mark 2 9 mm
- Docker remove container silent
- Polybius square decoder
